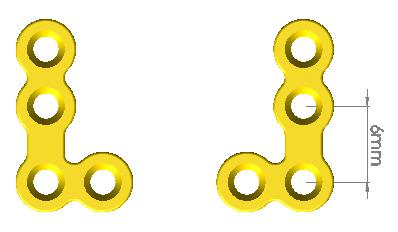
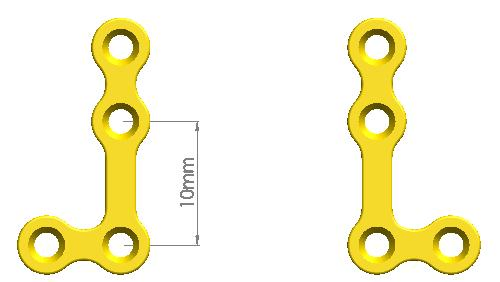
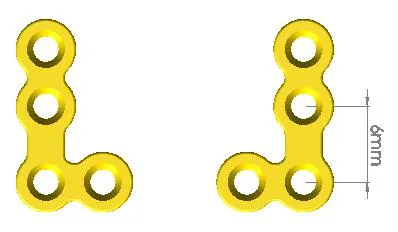
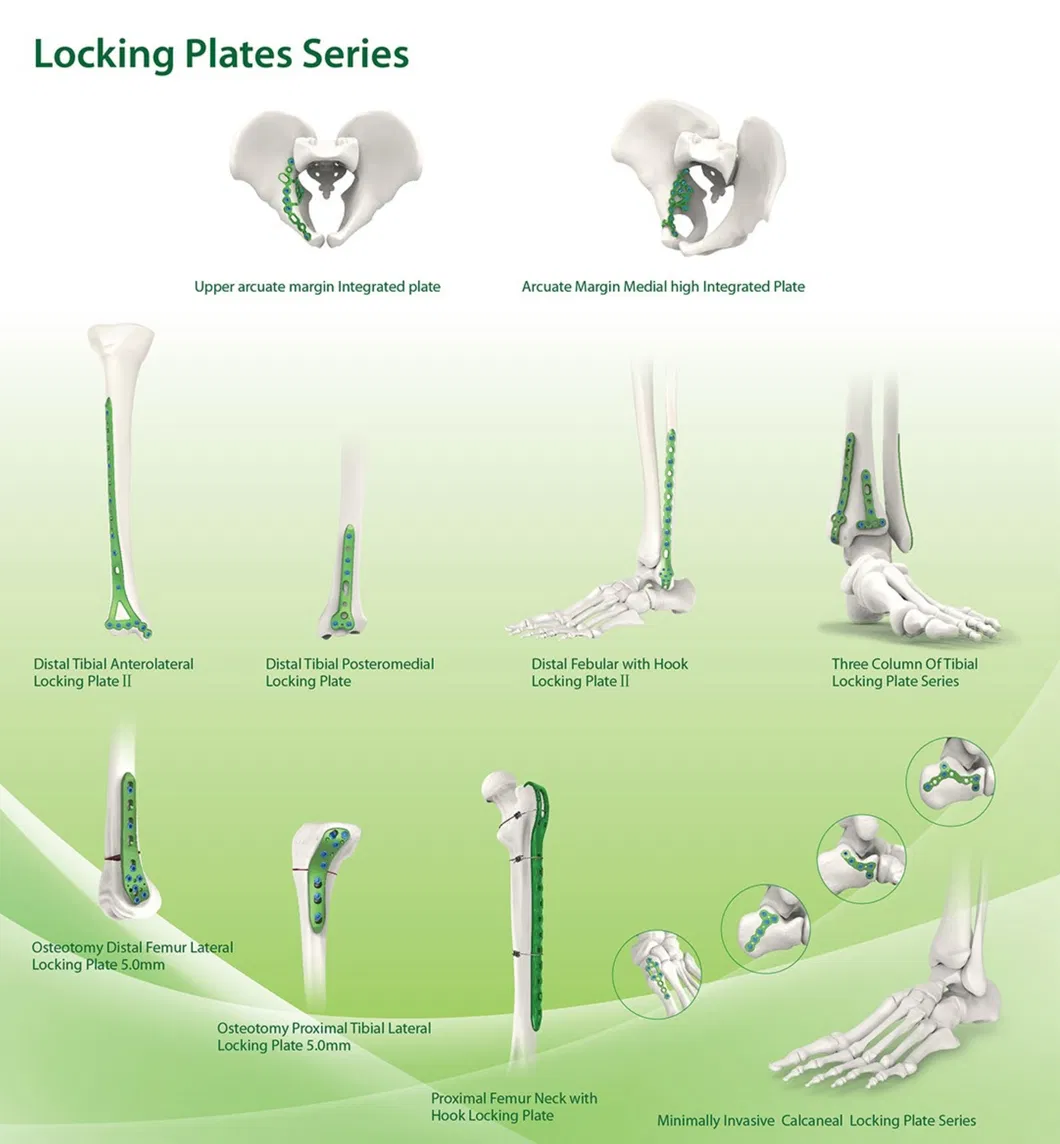
Company Profile
Established in 1958, we are engaged in the development, manufacture and sales of orthopedic implants and instruments. As a director company of the Medical Apparatus Industry Association, we focus on spinal pedicle screws, interlocking nails, locking plates, trauma screws, and surgical instruments.
Our products feature multi-varieties and complete specifications, having passed the approval of national and international superintendent offices. All products carry material codes and laser anti-fake marks for quality traceability.
Equipped with advanced CNC machines and a 100,000 Grade axenic purification chamber, we ensure top-tier quality. We were among the first to pass ISO 9001 certification and have won numerous technology awards for our innovative designs.

Frequently Asked Questions
What materials are used in your implants?
We primarily use high-grade Titanium for our maxillofacial plates and orthopedic implants to ensure biocompatibility and durability.
How do you guarantee the quality of your products?
Our products are produced according to EU CE standards. We conduct pre-production sample testing and a strict final inspection before shipment.
What is your monthly production capacity?
Our facility has a production capacity of approximately 10,000 pieces per month.
Do you offer OEM services?
Yes, we provide OEM manufacturing services for orthopedic implants and surgical instruments.
What payment methods are accepted?
We accept various payment terms including T/T (30% in advance), L/C, Western Union, and Cash.
What are your main product lines?
Our main products include metal bone plates, screw systems, interlocking intramedullary nails, spinal fixation devices, and external fixation tools.